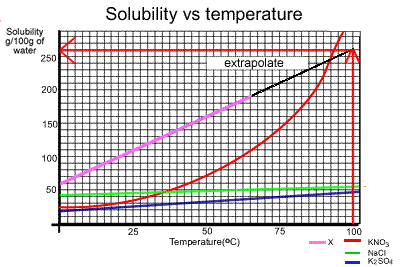
The amount of substance "X" that can dissolve in 100oC
is 260 grams
Hide
the solution
The amount of substance "X" that can dissolve in 100oC
is 260 grams

Hide
the solution
Hide
the solution
From the graph, we see that at 75oC we can dissolve 140 grams
of potassium nitrate in 100 grams of water. In 2 litres or 2,000 grams of
water we should be able to dissolve an amount equal to
140 X 20 = 2800 grams
|
Interpreting
data
exercise-2 |
|
The graph below
shows how the solubilities of 4 substances are affected by changes in
temperature. From the graph we can see that at 50oC approximately
70 grams of potassium nitrate (KNO3) will dissolve in 100
grams of water. |
 |
|
1) What is the maximum amount, in grams, of substance "X" that can dissolve in 100 grams of water at 50oC? 2) What is the maximum amount, in grams, of substance "X" that can dissolve in 100 grams of water at 100oC? Solution 3) Irene
wanted to see how much of each substance she could dissolve in two litres
of water at 50oC.
She listed the substances in order of greatest mass to the least mass
dissolved. 4) Stephen
dissolved potassium nitrate into 2 litres (2,000 grams) of water at
75oC until
no more could dissolve. He presented the solution to Jonathon and asked
him to guess the mass of potassium nitrate dissolved in 2 litres of
water. 5) Try this advanced problem. Irene added substance "X" to 100 grams of water at 50oC until no more could dissolve. She left the solution on a bench to cool. She returned after a few hours and noticed that the solution had cooled to 20oC and solid substance "X" was deposited on the bottom of the container. She filtered the solid substance and weighed it. The mass of substance "X" that was filtered was .Solution |